CALIBRATION AND THE ROLE OF CALIBRATION SAMPLES IN METAL OPTICAL EMISSION SPECTROMETER.
THEORY OF CALIBRATION:
 |
| Concentration vs Intensity calibration curve |
Calibration comprises measurement of calibration samples and determination of the functional relationship between the intensity ‘I’ of the line of an analyte and its concentration c in these samples. The functional relationship is the calibration function or calibration curve. It includes relationships between vaporisation, excitation, radiation offtake, dispersion and the measured value. Since spectrochemical analysis is a process of analysis is a process of analysis by comparison ( in contrast to absolute methods such as weighing ), it is necessary to carry out calibration with samples of accurately known concentration, the calibration samples.
The calibration function must not be confused with the function inverse to it-the read out or evaluation function. In the case of the calibration function I = f1 (c), the concentrations of the calibration samples are assumed to be free of error, and the errors (deviations from a best fit curve after correction of the intensities for systematic errors) are imputed entirely to the spectrometer method, so that the preconditions for regression calculations showing correlation coefficients as a quality index are useless. With the evaluation function c = f2 = ( I ) the concentration c of an analyte in an analytical sample is determined, which is accordingly subject to error, f2 = 1/f1.
For optical emission spectrometry there is no theory of calibration curves which can be used for practical purposes. There are formulae for which it is assumed that it is possible to represent the relationship between line intensity and concentration as a power function : I = I0 ck. The calibration function can be represented mathematically in various ways :
linear calibration function : I = f(c) = a0 + a1 c
non-linear calibration function : I =f(c) = a0 + a1 c +a2 c2+...+an cn
The extent to which the regression approaches the true course of the calibration
curve can be discerned from the residual scatter, namely at the point when the
addition of further terms to the approximation function does not produce any
further improvement in the residual scatter.
CALIBRATION SAMPLES
Fundamental role of the calibration samples is attested by international community and by International Standardisation Organization (ISO), which delivered the following definitions :
Reference Materials (RM) : they are Materials or substances whose properties are so well defined that they can be used to calibrate the instrument, verify the measure or assign values to the materials.
 |
| CRM sample with Spark analysis spots |
Certified Reference Materials (CRM) : they are Materials whose values concerning one or more properties are certified by means of a valid technical procedure and equipped by a Certificate or other documents from a qualified technical Body ( public or private Organization or Society., which deliver a certificate for the Reference Material )
Calibration samples present three disadvantages :
1) They are expensive
2) Their dimensions and shapes are not always available for the sample-holder stand of the spectrometer.
3) They are available only for some elements and concentrations
In some cases calibration samples can be synthesised, for example by alloying or diluting part of a charge. Because of this manipulation, the calculated values are rarely reliable and their composition should be confirmed by chemical analysis.
RECALIBRATION SAMPLES
When calibrating spectrometers with calibration samples (reference samples)
Recalibration samples are measured a number of times in order to obtain a reliable nominal value suitable for calibration. The additive and/or multiplicative changes in the sensitivity of the spectrometer bring about displacements of the calibration curves in the linear scale of the co-ordinate system. In order to trace (calculate) the actual intensity values at any later time back to the nominal intensity values submitted at the time of calibration a low (LP) and a high (HP) intensity is required for each analyte channel. In metal analysis with spark discharge the low points of all the analyte channels are usually measured with the pure base (Fe, Al, Cu,...). The high points are usually measured from synthetic samples having as many elements as possible with good homogeneity and precision.
The synthetic composition is given as a guide analysis and the samples often do not lie on the calibration curves. Mathematical procedure of calibration is a automated process.
In emission spectrometry recalibration samples run out, because of the polishing of the surface before recalibration. When recalibration samples are replaced there is no guarantee that, even with the same sample number, the new sample concentrations will correspond exactly to the sample being replaced. For this reason when calibrating a spectrometer for metal analysis, a minimum supply of recalibration samples should be available, for example five recalibration samples for each type.
The frequency of recalibration depends on the instrument and its use.
Interdependence with the instrument means that devices of the same kind, specially because of different phototubes stability, must be recalibrated at different intervals. Interdependence with use means that, even if stability is the same, recalibration frequency depends on the kind of analysis (traces analysis, sorting analysis).
(Note: The above post is written in context to calibration of Spark Optical emission spectrometer for metal and alloy analysis.)
ATOMIC FORCE MICROSCOPY
Atomic force microscopy (AFM) or scanning force microscopy (SFM) is a very high-resolution type of scanning probe microscopy, with demonstrated resolution on the order of fractions of a nanometer, AFM provides a 3D profile of the surface on a nanoscale, by measuring forces between a sharp probe (<10 nm) and surface at very short distance (0.2-10 nm probe-sample separation). The probe is supported on a flexible cantilever. The AFM tip “gently” touches the surface and records the small force between the probe and the surface.
The AFM measures the forces acting between a fine tip and a sample. The tip is attached to the free end of a cantilever, The cantilever is typically silicon or silicon nitride with a tip radius of curvature on the order of nanometres and it is brought very close to a surface. Attractive or repulsive forces resulting from interactions between the tip and the surface will cause a positive or negative bending of the cantilever. The bending is detected by means of a laser beam, which is reflected from the back side of the cantilever. The figure shows the basic concept of AFM :
What types of forces are measured?
 |
| Plot of force as a function of probe-sample separation |
The dominant interactions at short probe-sample distances in the AFM are Van der Waals interactions. However long-range interactions (i.e. capillary, electrostatic, magnetic) are significant further away from the surface. These are important in other SPM methods of analysis. During contact with the sample, the probe predominately experiences repulsive Van der Waals forces (contact mode). This leads to the tip deflection described previously. As the tip moves further away from the surface attractive Van der Waals forces are dominant (non-contact mode).
Modes of operation
CONTACT MODE
In the So-called contact-AFM mode, the tip makes soft “physical contact” with the surface of the sample. The deflection of the cantilever Dx is proportional to the force acting on the tip, via Hook’s law, F=-k. x, where k is the spring constant of the cantilever. In contact-mode the tip either scans at a constant small height above the surface or under the conditions of a constant force. In the constant height mode the height of the tip is fixed, whereas in the constant-force mode the deflection of the cantilever is fixed and the motion of the scanner in z-direction is recorded. By using contact-mode AFM, even “atomic resolution” images are obtained. For contact mode AFM imaging, it is necessary to have a cantilever which is soft enough to be deflected by very small forces and has a high enough resonant frequency to not be susceptible to vibration instabilities. Silicon Nitride tips are
used for contact mode. In these tips, there are 4 cantilever with different geometries attached to each substrate, resulting in 4 different spring constants.
 |
| Probe with four different cantilevers with different spring constants (N/m |
Advantages:
- High scan speeds
- Atomic resolution” is possible
- Easier scanning of rough samples with extreme changes in vertical topography
Disadvantages:
- Lateral forces can distort the image.
- Capillary forces from a fluid layer can cause large forces normal to the tip sample interaction
- Combination of these forces reduces spatial resolution and can cause damage to soft samples.
INTERMITTENT MODE (TAPPING)
The imaging is similar to contact. However, in this mode the cantilever is oscillated at its resonant frequency, Figure 4. The probe lightly “taps” on the sample surface during scanning, contacting the surface at the bottom of its swing. By maintaining a constant oscillation amplitude a constant tip-sample interaction is maintained and an image of the surface is obtained.
Advantages:
- Higher lateral resolution (1 nm to 5 nm).
- Lower forces and less damage to soft samples in air.
- Almost no lateral forces.
Disadvantage:
- Slower scan speed than in contact mode.
NON CONTACT MODE
(Attractive VdW) The probe does not contact the sample surface, but oscillates above the adsorbed fluid layer on the surface during scanning. (Note: all samples unless in a controlled UHV or environmental chamber have some liquid adsorbed on the surface). Using a feedback loop to monitor changes in the amplitude due to attractive VdW forces the surface topography can be measured.
Advantage:
- VERY low force exerted on the sample(10-12 N), extended probe lifetime.
Disadvantages:
- Lower lateral resolution, limited by tip-sample separation.
- Contaminant layer on surface can interfere with oscillation; usually need ultra-high vacuum (UHV) to have best imaging.
- Slower scan speed to avoid contact with fluid layer.
- Usually only applicable in extremely hydrophobic samples with a minimal fluid layer.
Applications :
The number of applications for AFM has exploded since it was invented in 1986 and Nowadays this technique is involved in many fields of Nanoscience and nanotechnology. The remarkable feature of STM and AFM instruments is their ability to examine samples not only in an ultrahigh vacuum but also on ambient conditions or even in liquids. AFM can image the non-conducting surfaces,
Possible applications of AFM are :
- Substrate roughness analysis.
- Step formation in thin film epitaxial deposition.
- Pinholes formation or other defects in oxides growth.
- Grain size analysis.
- Phase mode is very sensitive to variations in material properties, including surface stiffness, elasticity and adhesion.
- Comparing the tip-samples forces curves for materials to study the ratio of Young´s Modulus (graphite as a reference for measure of the indentation).
- Obtaining information of what is happening under indentation at very small loads .
- By In situ AFM analysis with changes in temperature we can study changes in the structure.
References :
1. Basic Theory Atomic Force Microscopy (AFM) by Robert A. Wilson and Heather A. Bullen,* Department of Chemistry, Northern Kentucky University, Highland Heights
2. Principles of atomic force microscopy (AFM) written by Arantxa Vilalta-Clemente , Aristotle University, Thessaloniki, Greece and Kathrin Gloystein, Aristotle University, Thessaloniki, Greece
GLOW DISCHARGE OPTICAL EMISSION SPECTROMETRY
GDOES made its first appearance in 1968 and was designed primarily for bulk spectrochemical analysis of various metals and their alloys. Since its introduction, this method has been steadily developed and has excelled in the areas of surface and coating analysis as well. Compared with conventional excitation techniques, the striking feature of Glow Discharge Technology is the ability to discern defined surface layers of the material being examined and analyze their chemical composition. In the field of metal analysis GDOES is ideal for concentration profile analysis and surface analysis. All kinds of surface treatment processes as well as surface coating processes can be monitored by analyzing the surface and near-surface areas the treated material. Coating thickness and chemical composition can be accurately measured using the technique of depth profile analysis. GDOES is the preferred method of analysis for materials that were previously impossible to analyze by traditional methods, and it is one of the fastest methods available.A stream of argon ions mill material from the sample surface. The sputtered material is then excited in a low pressure plasma discharge and resulting light emission is used to characterize and quantify the sample's composition. Glow Discharge offers an improved excitation source for fast, economical, accurate, and reliable sample turnaround. This source ultimately removes material from the sample surface which reduces the effects of metallurgical and chemical history inherent in all samples.
.
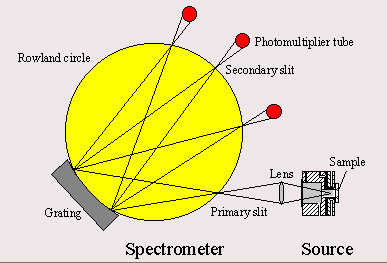 A Glow Discharge Optical Emission Spectrometer (GD-OES) is built of a glow discharge source and one or more optical spectrometers, including detectors, either Photomultiplier tubes or solid state detectors, usually CCD's. A schematic layout is given to the above. The spectrometer displayed here using a concave grating in the Rowland circle or Paschen-Runge configuration and photomultiplier tubes for the light detection.
A Glow Discharge Optical Emission Spectrometer (GD-OES) is built of a glow discharge source and one or more optical spectrometers, including detectors, either Photomultiplier tubes or solid state detectors, usually CCD's. A schematic layout is given to the above. The spectrometer displayed here using a concave grating in the Rowland circle or Paschen-Runge configuration and photomultiplier tubes for the light detection.
The use of solid state detectors, CCD's and photo diode array's have become a common alternative to Photomultiplier tubes. These detectors allow the acquisition of the entire spectrum, or at least a large portion of it, but are usually slower than Photomultiplier tubes and therefore not suitable for very short acquisition times used in thin film analysis.
The principle of operation is fairly easy to understand. In a glow discharge, cathodic sputtering is used to remove material layer by layer from the sample surface. The atoms removed from the migrate into the plasma where they are excited through collisions with electrons or metastable carrier gas atoms. The characteristic spectrum emitted by this excited atom is measured by the spectrometer.
GDOES can be used in many industries such as :
- Automotive industry and its suppliers.
- Metalworking industry.
- Iron and steel industry.
- Aerospace industry.
- Electronics industry.
- Glass and ceramics industry.
- Surface technology.
- Galvanizing industry.
- Photovoltaic industry.
- Scientific institutes.
Advantages of GDOES
- Limited matrix effect.
- Linear working curves.
- Minimal spectral interferences.
- Excellent precision.
- Analysis of difficult materials (as-cast iron, low melting point alloys).
- Automatic cleaning between samples.
- Low reference material and gas consumption.
OPTICAL EMISSION SPECTROSCOPY
Optical Emission Spectrometry (OES) is a fast, accurate and reliable method for quantitative analysis of metals to about 1–3% relative precision.Optical emission spectroscopy methods are among the most useful and flexible means of performing elemental analysis of Steel ,Copper , Aluminium,Magnesium, Lead etc and its alloys. Accurate analysis of metals and alloys requires that :
• The melt or bulk sample is homogeneous;
• Samples are representative of the melt or bulk sample and
• The metallurgical structure of the sample is similar to that of the standards used. The sample requirements are met by chill-cast disk samples collected as described in ASTM E 716-94 (or equivalents AS2612-83 or JISH 1305-1976). For the point-to-plane method of spectrochemical analysis , samples are machined on a lathe to give a smooth, clean surface just prior to analysis.
The sample is mounted in the spectrometer and a spark is generated between the sample and a tungsten electrode. The material is volatilized from the surface of the sample resulting in the emission of light. This light is diffracted into its component wavelengths inside the spectrometer. Fixed detectors simultaneously measure light intensity at wavelengths characteristic of each element. OES requires calibration of the spectrometer
with certified chill-cast standards, which must have the same matrix as the alloys to be tested. Therefore quantitative OES can only be performed on chillcast samples collected from the melt according to standard methods. Other techniques, such as Induction Coupled Plasma (ICP), can be used for quantitative analysis of alloys in other forms, for example billet slices, ingots, extrusions and forgings.
OES PRINCIPLE
The OES system consists of four main parts :
• Sample stand.
• Spark generator.
• Optics assembly.
• Data Acquisition Electronics assembly.
The clean sample is mounted in the stand and a spark is generated between the sample and a tungsten electrode. An example of this set up is illustrated in Figure 1. High purity argon is used as a discharge atmosphere to prevent any interaction between the atmosphere and the sample surface.
The material is violated from the surface of the sample resulting in the emission of light. When atoms are excited by an external energy source, some electrons move into higher energy levels. As these electrons drop back to their original levels, the atoms emit light (photons) of characteristic wavelength. The spectrometer collects the light emitted from the sample and splits it into its component wavelengths using a diffraction grating. Fixed detectors (photo multiplier tubes in above figure) simultaneously measure light intensity at wavelengths characteristic of each element. This is illustrated in Figure above. The intensity of the signals depends on the number of photons produced per unit time. The spectrometer is programmed for fixed sample types, elements and concentration ranges and only gives results within the limits of calibration


Nice blog. Everyone have to know the importance of steel. The best Steel Dealers will support us to grow. The quality is the main thing to construct a building. The quality will satisfy the customer needs.
ReplyDeleteAwesome Blog. Thanks for sharing useful content for us Atomic Force Microscope
ReplyDeleteA nice article here with some useful tips for those who are not used-to comment that frequently. Thanks for this helpful information I agree with all points you have given to us. I will follow all of them.
ReplyDeleteMetals Trading Brokers In India
Best Brokers For Commodity Trading In India
Thanks so much for a great post. I'd like to know more about these topics and hope that I can receive more insight into this topic.
ReplyDeleteFor more information visit here: Atomic Force Microscope
Nice post, thanks for sharing this post regarding ambient temperature Preservative with us.
ReplyDeleteNice Post.. Krishna steel and alloys is one of the leading and best fastener, Duplex steel fitting, Carbon steel flange manufacturer and supplier in India.
ReplyDeleteBelow are one of them:
Duplex steel fitting manufacturer in India,
Carbon steel forge fitting manufacturer in India
Carbon steel flange manufacturer in India
Butt weld Pipe Fitting Manufacturer in India
Stainless steel flange manufacturer in India
Structurals suppliers In India
Impressive and powerful suggestion by the author of this blog are really helpful to me. Carbon Steel Flange
ReplyDeleteWe read your blog , share most useful information in blog . carbon steel flanges
ReplyDeleteWe read your blog website, share most practical information in blog. Aluminium Supplier In Singapore
ReplyDeleteThis is actually good to read content of this blog. A is very general and huge knowledgeable platform has been known by this blog. I in reality appreciate this blog to have such kind of educational knowledge.
ReplyDeleteMonel 400 Plate
Share great information about your blog , Blog really helpful for us .
ReplyDelete316 Stainless Steel Pipe
Great blog ! I am impressed with suggestions of author.
ReplyDeleteStainless Steel Seamless Pipe
Your blog is awfully appealing. I am contented with your post. Monel 400 Plate
ReplyDeleteWith over 80 million people in America with poor credit, the credit repair industry is growing in leaps and bounds. Many consumers recognize that in order to take advantage of all the opportunities allotted to Americans, a favorable credit rating is a necessity. Once you've established that you need to remove incorrect, obsolete and/or unverifiable derogatory information from your credit report, now you must decide whether you will do it yourself or use a credit repair company. Stainless Braided Hose
ReplyDelete